15 results
Clinical factors and diagnoses associated with inappropriate urine-culture ordering in primary care
- Marissa Valentine-King, Barbara Trautner, Roger Zoorob, Michael Hansen, Jennifer Matas, Robert Atmar, Larissa Grigoryan
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, p. s1
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Inappropriate urine-culture ordering is associated with increased antibiotic prescribing in myriad care environments, including acute and long-term care. In primary care, where urinary tract infections (UTIs) are commonly encountered, the appropriateness of urine-culture ordering has not been well described. We examined the appropriateness of urine-culture ordering and factors associated with inappropriate urine-culture ordering in primary care. Methods: We conducted a secondary analysis of data from a previous prospective study that included patients aged ≥18 years presenting with provider-suspected UTI with an accompanying urine culture at 2 safety-net, primary-care clinics in Houston, Texas, between November 2018 and March 2020. Patients with complicated or uncomplicated UTI were included, but those with a urinary catheter and pregnant females were excluded. Urine cultures were considered appropriate if the patient had an evidence-based symptom of UTI (ie, dysuria, frequency, urgency, hematuria, fever, chills, costovertebral angle tenderness, suprapubic, pelvic, or flank pain, or nephrolithiasis) as a diagnostic code or listed in providers’ free-text documentation. Diagnostic codes for symptoms that were not evidence based were grouped into categories based on body system, visit type (eg, routine visit), or sign or symptom clusters. We evaluated the relationships among demographic and clinical factors, the clinic visited, and non–evidence-based diagnostic codes with inappropriately ordered cultures. Results: We examined 870 cultures from 807 patients. Overall, 61.5% of patients were Hispanic (61.5%) and 23% were African American or Black. Also, 70.6% were women; the mean age was 49.2 years (SD, 14.6); and the mean Elixhauser score was 1.9 (SD, 5.4). Among the 870 cultures, 210 (24%) were ordered inappropriately. Dysuria (n = 289), frequency (n = 129), and UTI or cystitis (n = 117) were the most common, evidence-based codes among appropriate cultures. In the adjusted model, the nonteaching clinic (aOR, 6.33) and diagnostic codes comprising the following categories were associated with inappropriate culturing: acute lower back pain (aOR, 5.42), cardiac-related visits (aOR, 2.41), urinary incontinence (aOR, 4.46), routine health visits (aOR, 3.66), urine characteristics (aOR, 14.32), voiding difficulties (aOR, 3.88), and well-woman visits with a gynecological exam or family planning aspect (aOR, 12.27) (all P < .05). Conclusions: This research highlights potential gaps or miscues in provider behavior related to urine culture ordering, and unveiled problematic culturing related to urine characteristics and to routine visits, especially of a gynecological nature. This information can be incorporated into diagnostic stewardship interventions to address misconceptions, and to further explore the reasoning or processes wherein urine cultures are ordered for routine visits.
Financial support: NIAID UM1AI104681
Disclosure: None
Identifying nonprescription antibiotic users with screening questions in a primary care setting
- Eva Amenta, Marissa Valentine-King, Lindsey Laytner, Michael Paasche-Orlow, Richard Street, Kenneth Barning, Thomas Porter, Hammad Mahmood, Barbara Trautner, Larissa Grigoryan
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s20-s21
-
- Article
-
- You have access Access
- Open access
- Export citation
-
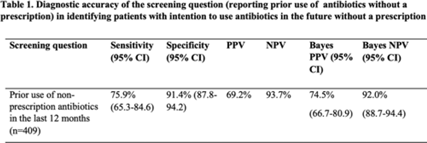
Background: Antibiotic use without a prescription (nonprescription use) leads to antibiotic overuse, with negative consequences for patient and public health. We studied whether screening patients for prior nonprescription antibiotic use in the past 12 months predicted their intentions to use them in the future. Methods: A survey asking respondents about prior and intended nonprescription antibiotic use was performed between January 2020 and June 2021 among patients in waiting rooms of 6 public clinics and 2 private emergency departments in economically and socially diverse urban and suburban areas. Respondents were classified as prior nonprescription users if they reported previously taking oral antibiotics without contacting a doctor, dentist, or nurse. Intended use was defined as answering “yes” or “maybe” to the question, “Would you use antibiotics without contacting a doctor, nurse, or dentist?” We calculated the sensitivity, specificity, and positive and negative predictive value (PPV and NPV) of prior nonprescription antibiotic use in the past 12 months for future intended nonprescription use. Bayes PPV and NPV were also calculated, considering the prevalence of nonprescription antibiotic use (24.8%) in our study. Results: Of the 564 patients surveyed, the median age was 51 years (SD, 19–92), with 72% of patients identifying as female. Most were from the public healthcare system (72.5%). Most respondents identified as Hispanic or Latino(a) (47%) or African American (33%), and 57% received Medicaid or the county financial assistance program. Prior nonprescription use was reported by 246 (43%) of 564 individuals, with 91 (16%) reporting nonprescription use within the previous 12 months. Intention to use nonprescription antibiotics was reported by 140 participants (25%). The sensitivity and specificity of prior nonprescription use in the past 12 months to predict the intention to use nonprescription antibiotics in the future were 75.9% (95% CI, 65.3–84.6) and 91.4% (95% CI, 87.8–94.2), respectively. After the Bayes’ adjustment, the PPV and NPV of prior use to predict future intention were 74.5% (95% CI, 66.7–80.9) and 92.0% (95% CI, 88.7–94.4) (Table 1). Conclusions: These results show that prior nonprescription antibiotic use in the past 12 months predicted the intention to use nonprescription antibiotics in the future (PPV of 75%). As a stewardship effort, we suggest clinicians use a simple question about prior nonprescription antibiotic use in primary-care settings as a screening question for patients at high risk for future nonprescription antibiotic use.
Financial support: HSQR-R 5R01HS026901-04
Disclosure: None
Perspectives on nonprescription antibiotic use among Hispanic patients in the Houston metroplex: A qualitative study
- Lindsey Laytner, Patricia Chen, Susan Nash, Michael Paasche-Orlow, Kiara Olmeda, Juanita Salinas, Richard Street, Roger Zoorob, Barbara Trautner, Larissa Grigoryan
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, p. s20
-
- Article
-
- You have access Access
- Open access
- Export citation
-
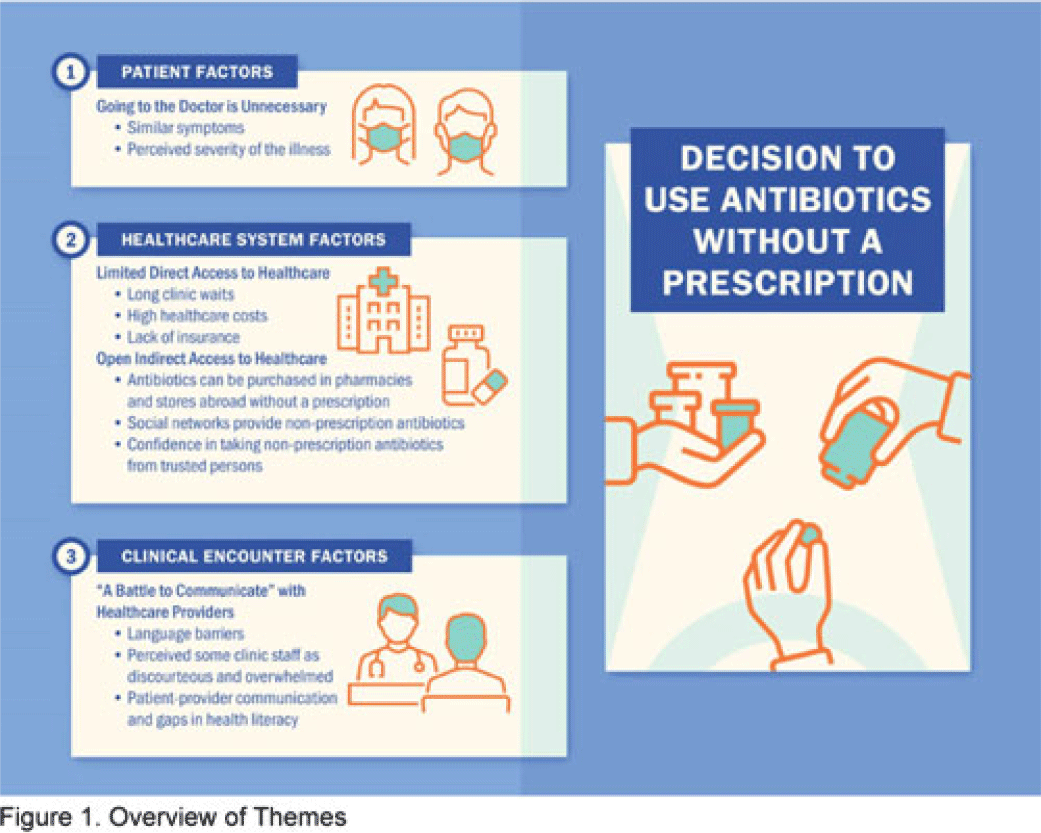
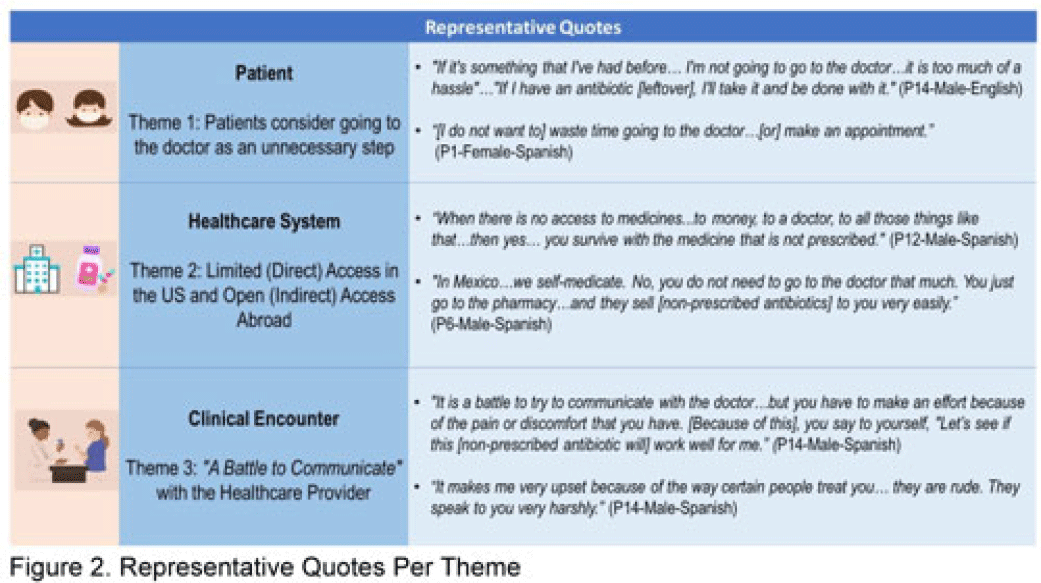
Background: Nonprescription antibiotic use includes taking an antibiotic without medical guidance (eg, leftover antibiotics, antibiotics from friends or relatives, or antibiotics purchased without a prescription). Nonprescription use contributes to antimicrobial resistance, adverse drug reactions, interactions, and superinfections such as Clostridioides difficile colitis. Qualitative studies exploring perspectives regarding nonprescription use among Hispanic patients are lacking. We used the Kilbourne Framework for Advancing Health Disparities Research to identify factors influencing Hispanic patients’ nonprescription use and to organize our findings. Methods: Our study includes Hispanic primary-care clinic patients with different types of health coverage in the Houston metroplex who endorsed nonprescription use in a previous quantitative survey. Semistructured interviews explored the factors promoting nonprescription use in Hispanic adults. Interviews were conducted remotely, in English or Spanish, between May 2020 and October 2021. We used inductive coding and thematic analysis to identify the factors and motives for nonprescription use. Results: Of the 35 Hispanic participants surveyed, 69% were female and between the ages of 27 and 66. All participants had some form of healthcare coverage (eg, Medicare or private insurance, Medicaid, or the county financial assistance program). Participants reported obtaining antibiotics from their own leftover prescriptions and through trusted persons (eg, herbalists, pharmacists, friends/relatives, and others), buying them under the counter in US markets, and purchasing them without a prescription outside the United States. Thematic analysis revealed the factors contributing to nonprescription use (Fig. 1). Themes included beliefs that the ‘doctor visit was unnecessary,’ ‘limited direct access to healthcare’ in the United States (due to limited insurance coverage, high costs of the doctor’s visits and medications, and long clinic wait times), ‘more open indirect access to healthcare’ abroad and under the counter in the United States, and communication difficulties (eg, language barriers with clinicians, perceived staff rudeness, and gaps in health literacy). Figure 2 shows representative quotes across thematic domains. Participants expressed having confidence in medical recommendations from pharmacists and trusted community members in their social networks. Conclusions: Antibiotic stewardship interventions that include pharmacist-driven patient education regarding appropriate antibiotic use may decrease nonprescription antibiotic use in Hispanic communities. Additionally, improving access to care while addressing communication barriers and cultural competency in clinics may improve primary care delivery and reduce potentially unsafe antibiotic use.
Disclosure: None
Quantifying the Implementation and Cost of a Multisite Antibiotic Stewardship Intervention for Asymptomatic Bacteriuria
- Eva Amenta, Larissa Grigoryan, Suja S. Rajan, David Ramsey, Jennifer R. Kramer, Annette Walder, Andrew Chou, John N. Van, Sarah L. Krein, Sylvia Hysong, Aanand D. Naik, Barbara W. Trautner
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue 1 / 2023
- Published online by Cambridge University Press:
- 30 June 2023, e115
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Objective:
The intensity of an antibiotic stewardship intervention to achieve clinical impact is not known. We conducted a multisite dissemination project of an intervention to reduce treatment of asymptomatic bacteriuria (ASB) and studied: (1) the association between implementation metrics and clinical outcomes and (2) the cost of implementation.
Design/Setting/Participants:A central site facilitated a multimodality intervention to decrease unnecessary urine cultures and antibiotic treatment in patients with ASB at 4 Veterans Affairs medical centers.
Methods:The intervention consisted of a decision support aid algorithm and interactive teaching cases that provided in the moment audit and feedback on how to manage ASB. Implementation outcomes included minutes spent in intervention delivery, number of healthcare professionals reached, and number of sessions delivered. Clinical outcomes included days of antibiotic therapy (DOT), length of antibiotic therapy (LOT), and number of urine cultures ordered per 1000 bed days. Personnel reported weekly time logs.
Results:Minutes spent in intervention delivery were inversely correlated with two clinical outcomes, DOT (R −0.3, P = .04) and LOT (R −0.3, P = .02). Number of healthcare professionals reached and number of sessions delivered were not correlated with clinical outcomes of DOT (R –0.003, P = .98, R = −0.059, P = .69) or LOT (R +0.073, P = .62, R −0.102, P = .49). Physician champions spent an average of 3.8% of effort on the intervention. The implementation cost was USD 22,299/year per site on average.
Conclusions:The amount of time local teams spent in delivery of an antibiotic stewardship intervention was correlated with the desired decrease in antibiotic use. Implementing this successful antibiotic stewardship intervention required minimal time.
Identifying symptoms/illnesses and situations that predispose outpatients to use antibiotics in two healthcare systems – CORRIGENDUM
- Lindsey Laytner, Barbara Trautner, Michael Hansen, Roger Zoorob, Osvaldo Alquicira, Juanita Salinas, Fareed M. Khan, Larissa Grigoryan
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue 1 / 2022
- Published online by Cambridge University Press:
- 04 October 2022, e164
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
Identifying symptoms/illnesses and situations that predispose outpatients to use antibiotics in two healthcare systems
- Lindsey Laytner, Larissa Grigoryan, Barbara Trautner, Osvaldo Alquicira, Juanita Salinas, Michael Hansen, Roger Zoorob, Fareed M. Khan
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, pp. s68-s69
-
- Article
-
- You have access Access
- Open access
- Export citation
-
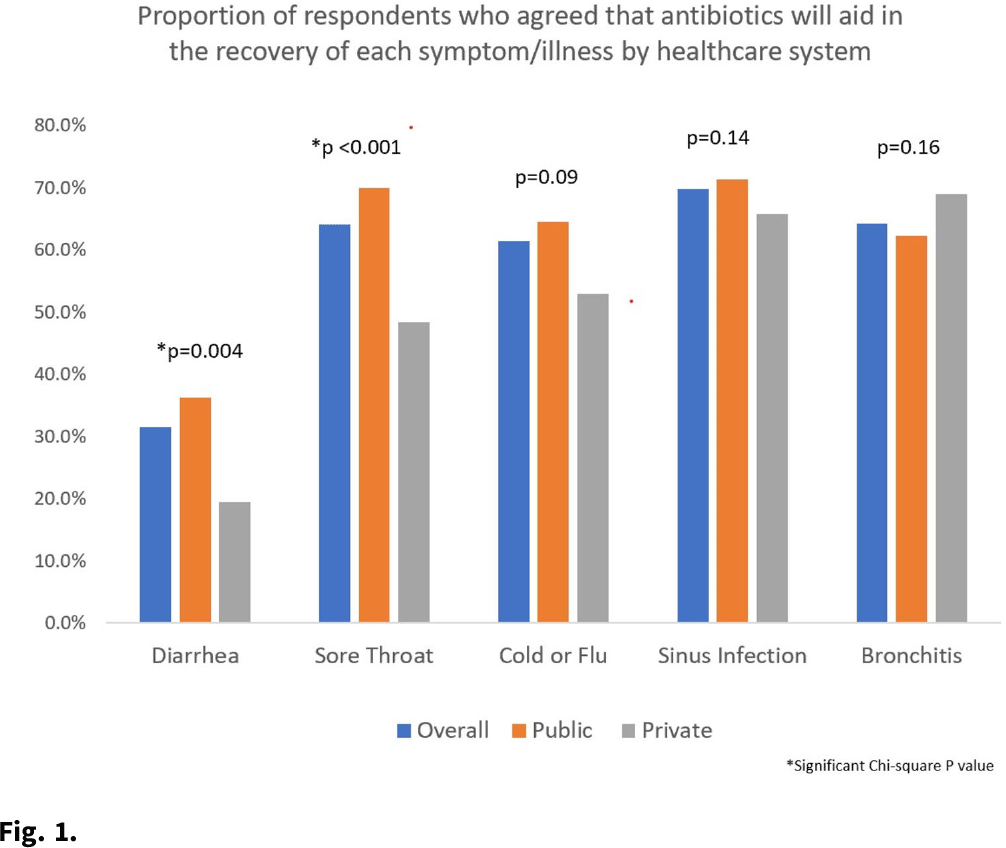
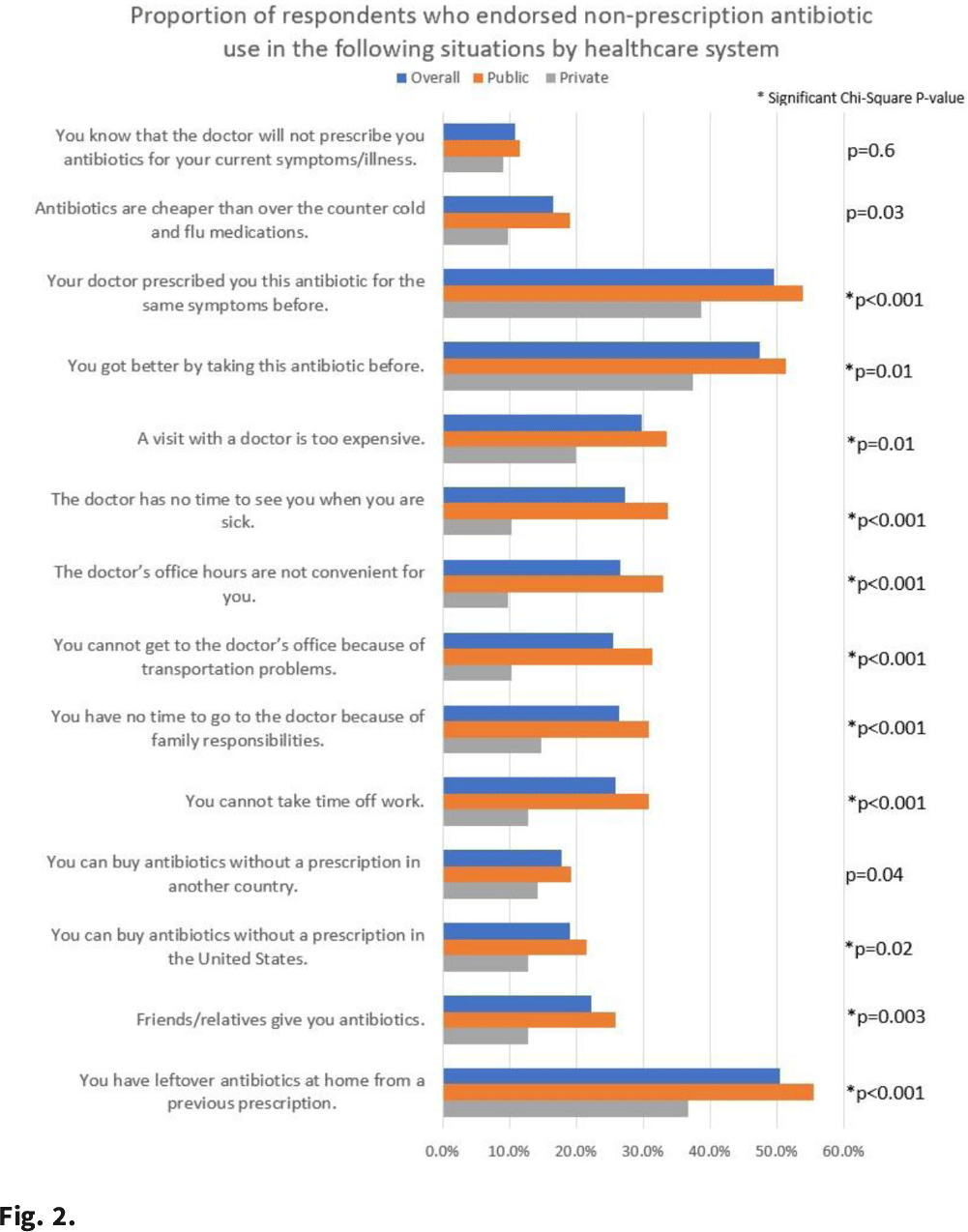
Background: Taking antibiotics outside the guidance of a clinician (nonprescription use) is a potential safety issue and runs counter to antibiotic stewardship efforts. We identified the symptoms and illnesses and situations that may predispose patients to take antibiotics, and we compared these findings between patients attending public primary care clinics and private emergency departments. Methods: A cross-sectional survey was conducted between January 2020 and March 2021 in 6 primary care clinics and 2 emergency departments in the United States. We queried patients about 5 symptoms and illnesses (Fig. 1) and 14 situations (Fig. 2) to investigate whether these would lead the patients to take antibiotics without a prescription. We used the χ2 test to compare the symptoms and illnesses and situations between the respondents from public and private healthcare systems. We set the P value for significance at <.025. Results: In total, the survey had 564 respondents (median age, 49.7 years; range, 19–92), and 72% were female. Most respondents identified as either Hispanic or Latina/Latino (46.6%) or African American or Black (33%), followed by White (15.8%), and other (4.6%). Most respondents had visited public clinics (72%). The most common insurance status for our respondents included Medicaid or county financial assistance program (56.6%), followed by private insurance or Medicare (36.7%) and self-pay (6.7%). In public primary care clinics, only 23% had private insurance or Medicare compared to 72.9% in private emergency departments. Of those surveyed, 69% agreed that antibiotics would improve the recovery from sinus infections, followed by bronchitis (64%), sore throat (64%), cold/flu (61.4%), and diarrhea (31.5%). The proportions of respondents who believed that antibiotics would improve the recovery from diarrhea (36.2% vs 19.4%; P = .004) and sore throat (59.9% vs 48.4%; P < .001) were significantly higher among public versus private outpatient respondents. We did not find significant differences for cold/flu, sinus infection, or bronchitis between these 2 healthcare systems (Fig. 1). In 11 of the 14 situations, patients in public clinics were more likely to report a likelihood of using nonprescription antibiotics than the patients visiting the private emergency rooms (Fig. 2). Conclusions: Future stewardship interventions should be aware of the symptoms and illnesses and situations that may influence outpatients to take nonprescription antibiotics. Addressing modifiable factors (eg, leftover antibiotics, antibiotics given by friends or family, and antibiotics available without a prescription in stores or markets) may also curtail these unsafe practices and reduce antibiotic resistance.
Funding: None
Disclosures: None
Prior cultures predict subsequent susceptibility in patients with recurrent urinary tract infections
- Marissa Valentine-King, Barbara Trautner, Roger Zoorob, George Germanos, Jason Salemi, Kalpana Gupta, Larissa Grigoryan
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, p. s67
-
- Article
-
- You have access Access
- Open access
- Export citation
-
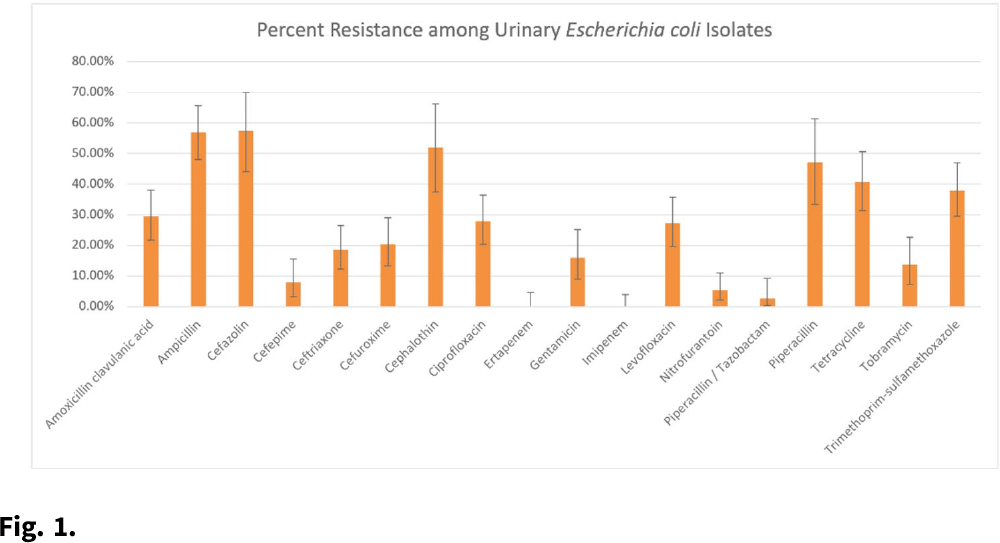
Background: Patients with recurrent urinary tract infections (rUTI) experience frequent exposure to antimicrobial regimens, leaving them at higher risk for developing antibiotic resistance. Little information on the prevalence of antibiotic resistance among patients with rUTI has been published. Although the IDSA recommends using a prior culture to guide empiric treatment, studies have not examined the predictive ability of a prior culture among patients meeting rUTI criteria. We constructed an antibiogram and evaluated test metrics, including sensitivity, specificity, and positive predictive value (PPV) and negative predictive values (NPV) of a prior culture (any organism), on predicting resistance (PPV) or susceptibility (NPV) of a future culture among patients with uncomplicated rUTI in an outpatient setting. Methods: We retrospectively extracted electronic health record data from outpatients aged ≥18 years who had an ICD-10 code for cystitis listed twice in 6 months or thrice in 12 months between November 1, 2016, and December 31, 2018. Patients sought care at either urology or primary care practices within an academic medical center in Houston, Texas. Patients with functional or structural abnormalities of the genitourinary tract, signs or symptoms of pyelonephritis, or pregnancy were excluded. Antibiogram data were reported for uropathogens with ≥30 isolates, and intermediate results were considered resistant. Test metrics and Bayes’ PPV and NPV were calculated using standard formulas. Results: We included 597 visits from 232 unique patients. Most were White (63%) and female (92%), and the cohort had a median age of 58 (IQR, 41–68). Among 310 rUTI episodes with a urine culture, 189 (61%) had at least 1 uropathogen isolated, and Escherichia coli (n = 130, 66%) was most common among all 196 uropathogens. E. coli isolates had >20% resistance to 10 of 18 antibiotics (Fig. 1). E. coli resistance to ciprofloxacin was 27.9%, resistance to nitrofurantoin was 5.5%, and resistance to trimethoprim-sulfamethoxazole was 38.0%. The PPVs for predicting resistance were highest for ceftriaxone (0.86; 95% CI, 0.60–0.96), ciprofloxacin (0.84; 95% CI, 0.63–0.94), and levofloxacin (0.84; 95% CI, 0.63–0.94). NPVs of resistance were highest for gentamicin (0.97; 95% CI, 0.83–1.00), ceftriaxone (0.94; 95% CI, 0.86–0.98), and cefepime (0.94; 95% CI, 0.84–0.98), whereas NPVs for cefuroxime, ciprofloxacin, levofloxacin, and nitrofurantoin were all >0.83. Conclusions: We detected considerable antibiotic resistance among patients with rUTI to commonly prescribed antibiotics. Prior urine culture susceptibility demonstrated moderate-to-high PPVs for predicting future resistance to ceftriaxone and fluoroquinolones as well as high NPVs for several cephalosporins and fluoroquinolones, which could inform empiric prescribing choices.
Funding: This investigator-initiated research study was funded by Rebiotix, a Ferring Company.
Disclosures: None
Analysis of recurrent urinary tract infection management in women seen in outpatient settings reveals opportunities for antibiotic stewardship interventions
- Marissa A. Valentine-King, Barbara W. Trautner, Roger J. Zoorob, George Germanos, Michael Hansen, Jason L. Salemi, Kalpana Gupta, Larissa Grigoryan
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue 1 / 2022
- Published online by Cambridge University Press:
- 17 January 2022, e8
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Objectives:
We characterized antibiotic prescribing patterns and management practices among recurrent urinary tract infection (rUTI) patients, and we identified factors associated with lack of guideline adherence to antibiotic choice, duration of treatment, and urine cultures obtained. We hypothesized that prior resistance to nitrofurantoin or trimethoprim–sulfamethoxazole (TMP-SMX), shorter intervals between rUTIs, and more frequent rUTIs would be associated with fluoroquinolone or β-lactam prescribing, or longer duration of therapy.
Methods:This study was a retrospective database study of adult women with International Classification of Diseases, Tenth Revision (ICD-10) cystitis codes meeting American Urological Association rUTI criteria at outpatient clinics within our academic medical center between 2016 and 2018. We excluded patients with ICD-10 codes indicative of complicated UTI or pyelonephritis. Generalized estimating equations were used for risk-factor analysis.
Results:Among 214 patients with 566 visits, 61.5% of prescriptions comprised first-line agents of nitrofurantoin (39.7%) and TMP-SMX (21.5%), followed by second-line choices of fluoroquinolones (27.2%) and β-lactams (11%). Most fluoroquinolone prescriptions (86.7%), TMP-SMX prescriptions (72.2%), and nitrofurantoin prescriptions (60.2%) exceeded the guideline-recommended duration. Approximately half of visits lacked a urine culture. Receiving care through urology via telephone was associated with receiving a β-lactam (adjusted odds ratio [aOR], 6.34; 95% confidence interval [CI], 2.58–15.56) or fluoroquinolone (OR, 2.28; 95% CI, 1.07–4.86). Having >2 rUTIs during the study period and seeking care from a urology practice (RR, 1.28, 95% CI, 1.15–1.44) were associated with longer antibiotic duration.
Conclusions:We found low guideline concordance for antibiotic choice, duration of therapy and cultures obtained among rUTI patients. These factors represent new targets for outpatient antibiotic stewardship interventions.
Analysis of Recurrent Urinary Tract Infection Management in Outpatient Settings Reveals Opportunities for Antibiotic Stewards
- Marissa Valentine-King, Barbara Trautner, Roger Zoorob, George Germanos, Jason Salemi, Kalpana Gupta, Larissa Grigoryan
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, p. s34
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Studies of antibiotic prescribing choice and duration have typically excluded women with recurrent UTI (rUTI), yet the Infectious Disease Society of America (IDSA) UTI treatment guidelines are applicable to recurrent and sporadic cystitis. We sought to better understand prescribing practices among uncomplicated rUTI patients in terms of choice of drug, duration of therapy, and the risk factors for receiving guideline-discordant therapy. Methods: We performed a retrospective database study by extracting electronic health record data from adults seen at academic primary care, internal medicine, or urology practices between November 2016 and December 2018. Inclusion criteria included having ≥2 or ≥3 International Classification of Diseases Tenth Edition (ICD-10) cystitis codes recorded within a 6- or 12-month period, respectively. We excluded patients with ICD-10 codes indicating any structural or functional genitourinary comorbidities, interstitial cystitis, vaginosis, compromised immune systems, or pregnancy in the prior year. Patients were also excluded if they had signs or symptoms of pyelonephritis at presentation. Results: Overall, 232 patients presented for 597 outpatient visits. Most were married (52.2%), non-Hispanic white (62.9%), and female (92.2%), with a median age of 58 years (IQR, 41–68). Only 21% of visits with an antibiotic prescribed for treatment consisted of a first-line therapy agent prescribed for the recommended duration. In terms of antibiotic choice, these agents were prescribed in 58.4% of scenarios, which primarily included nitrofurantoin (37.8%) and trimethoprim-sulfamethoxazole (TMP-SMX) (20.3%). Guideline-discordant choices of fluoroquinolones (28.8%), and β-lactams (11.2%) were the second and third most commonly prescribed drug categories, respectively. Multinomial logistic regression identified age (OR, 1.02; 95% CI, 1.002–1.04) or having a telephone visit (OR, 3.17; 95% CI, 1.54–6.52) as independent risk factors for receiving a β-lactam. The duration exceeded the 3-day guideline recommendation in 87.6% of fluoroquinolones and 73% of TMP-SMX (73%) prescriptions, and 61% of nitrofurantoin prescriptions exceeded the recommended 5-day duration. Multiple logistic regression analysis revealed that seeking care at a urology clinic (OR, 2.81; 95% CI, 1.59–5.17) served as an independent factor for therapy duration exceeding guideline recommendations. Conclusions: This retrospective study revealed shortcomings in prescribing practices in the type and duration of therapy for rUTI. rUTI as well as sporadic UTI are important targets for outpatient antibiotic stewardship interventions.
Funding: This investigator-initiated research study was funded by Rebiotix Inc, a Ferring Company.
Disclosures: None
Optimizing Urine Collection Represents an Important Stewardship Opportunity in Primary Care
- Larissa Grigoryan, Jennifer Matas, Michael Hansen, Samuel Willis, Lisa Danek, Anna Katta, Kenneth Muldrew, Mohammad Zare, Forrest Hudson, Robert Atmar, Andrew Chou, Barbara Trautner
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, pp. s14-s15
-
- Article
-
- You have access Access
- Open access
- Export citation
-
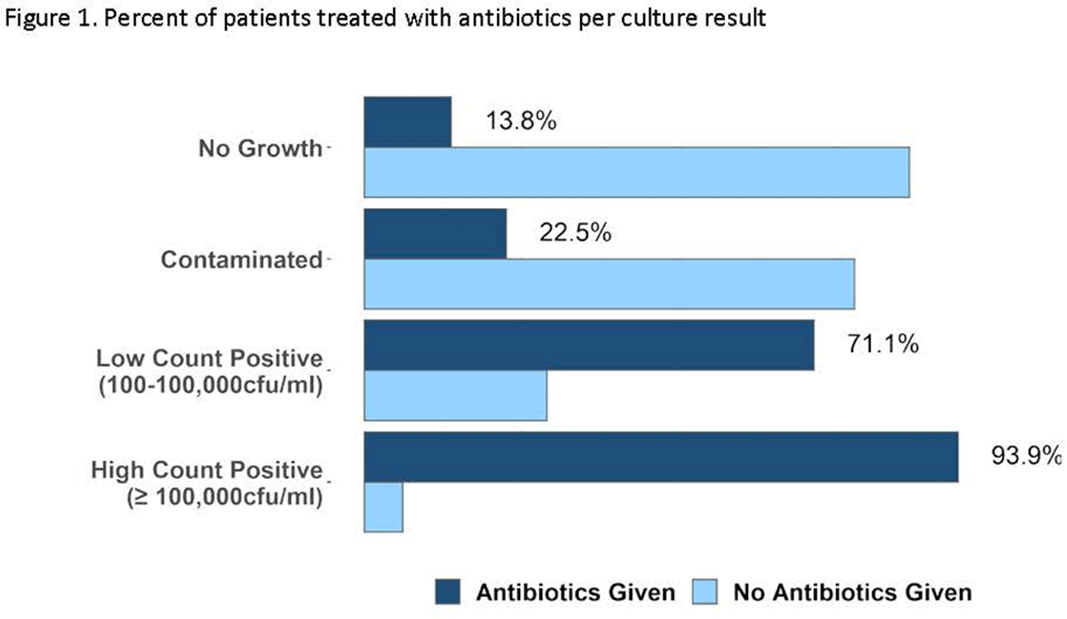
Background: Urine cultures are the most common microbiological tests in the outpatient setting and heavily influence treatment of suspected urinary tract infections (UTIs). Antibiotics for UTI are usually prescribed on an empiric basis in primary care before the urine culture results are available. However, culture results may be needed to confirm a UTI diagnosis and to verify that the correct antibiotic was prescribed. Although urine cultures are considered as the gold standard for diagnosis of UTI, cultures can easily become contaminated during collection. We determined the prevalence, predictors, and antibiotic use associated with contaminated urine cultures in 2 adult safety net primary care clinics. Methods: We conducted a retrospective chart review of visits with provider-suspected UTI in which a urine culture was ordered (November 2018–March 2020). Patient demographics, culture results, and prescription orders were captured for each visit. Culture results were defined as no culture growth, contaminated (ie, mixed flora, non-uropathogens, or ≥3 bacteria isolated on culture), low-count positive (growth between 100 and 100,000 CFU/mL), and high-count positive (>100,000 CFU/mL). A multivariable multinomial logistic regression model was used to identify factors associated with contaminated culture results. Results: There were 1,265 visits with urine cultures: 264 (20.9%) had no growth, 694 (54.9%) were contaminated, 159 (12.6%) were low counts, and 148 (11.7%) were high counts. Encounter-level factors are presented in Table 1. Female gender (adjusted odds ratio [aOR], 15.8; 95% confidence interval [CI], 10.21–23.46; P < .001), pregnancy (aOR, 13.98; 95% CI, 7.93–4.67; P < .001), and obesity (aOR, 1.9; 95% CI 1.31–2.77; P < .001) were independently associated with contaminated cultures. Of 264 patients whose urine cultures showed no growth, 36 (14%) were prescribed an antibiotic. Of 694 patients with contaminated cultures, 153 (22%) were prescribed an antibiotic (Figure 1). Conclusions: More than half of urine cultures were contaminated, and 1 in 5 patients were treated with antibiotics. Reduction of contamination should improve patient care by providing a more accurate record of the organism in the urine (if any) and its susceptibilities, which are relevant to managing future episodes of UTI in that patient. Optimizing urine collection represents a diagnostic stewardship opportunity in primary care.
Funding: This study was supported by the National Institute of Allergy and Infectious Diseases of the National Institutes of Health (grant no. UM1AI104681). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Disclosures: None
Table 1. 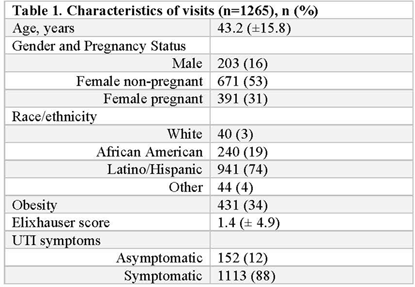
Figure 1.
Organizational Readiness to Change Assessment Highlights Differential Readiness for Antibiotic Stewardship
- Melanie Goebel, Barbara Trautner, Yiqun Wang, John Van, Laura Dillon, Payal Patel, Dimitri Drekonja, Christopher Graber, Paola Lichtenberger, Christian Helfrich, Larissa Grigoryan
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s492-s493
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: Targeted antibiotic stewardship interventions are needed to reduce unnecessary treatment of asymptomatic bacteriuria (ASB). Organizational readiness for change is a precursor to successful change implementation. The Organizational Readiness to Change Assessment (ORCA) is a validated survey instrument that has been used to detect potential obstacles and tailor interventions. In an outpatient stewardship study, primary care practices with high readiness to change trended toward greater improvements in antibiotic prescribing. We used the ORCA to assess barriers to change before implementing a multicenter inpatient stewardship intervention for ASB. Methods: Surveys were self-administered by healthcare professionals in inpatient medicine and long-term care units at 4 geographically diverse Veterans’ Affairs facilities during January–December 2018. Participants included providers (physicians, physician assistants, and nurse practitioners), nurses, pharmacists, infection preventionists, and quality managers. The survey included 7 subscales: evidence (perceived evidence strength) and 6 context subscales (favorability of the organizational context to support change). Responses were scored on a 5-point Likert scale, with 1 meaning very weak or strongly disagree. Scores were compared between professional types and sites. We also measured allocated employee effort for stewardship at each site. Results: Overall, 104 surveys were completed, with an overall response rate of 69.3%. For all sites combined, the evidence subscale had the highest score of the 7 subscales (mean, 4; SD, 0.9); the resources subscale was significantly lower than other subscales (mean, 2.8; SD, 0.9; P < .001). Scores for budget and staffing resources were lower than scores for training and facility resources (P < .001 for both comparisons). Pharmacists had lower scores than providers for the staff culture subscale (P = .04). Comparing subscales between sites, ORCA scores were significantly different for leadership behavior (communication and management), measurement (goal setting and accountability), and general resources (Fig. 1). The site with the lowest scores for resources (mean, 2.4) also had lower scores for leadership behavior and measurement, and lower pharmacist effort devoted to antibiotic stewardship. Conclusions: Although healthcare professionals endorsed the evidence about nontreatment of ASB, perceived barriers to antibiotic stewardship included inadequate resources and lack of leadership support. These findings provide targets for tailoring the intervention to maximize the success of our stewardship program. Our support to sites with lower leadership scores includes training of local champions who are dedicated to supporting the intervention. For sites with low scores for resources, our targeted implementation strategies include analyzing local needs and avoiding increased workload for existing personnel.
Funding: None
Disclosures: None
A Conceptual Framework for Understanding How and Why People Take Antibiotics Without a Prescription
- Larissa Grigoryan, Osvaldo Alquicira, Susan Nash, Melanie Goebel, Barbara Trautner
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, p. s93
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: The reported prevalence of nonprescription antibiotic use in the United States varies from 5% among socioeconomically and ethnically diverse primary care patients to 66% among Latino migrant workers. Reports indicate that people obtain and take antibiotics from stores or flea markets in the United States, friends or relatives, and leftover antibiotics from previous prescriptions. This unsafe practice may lead to unnecessary and inappropriate antibiotic use and increases the risk of antibiotic resistance. As groundwork to develop an intervention to decrease nonprescription antibiotic use, we mapped reported drivers of nonprescription use to the Kilbourne conceptual framework for advancing health disparities research. Methods: The Kilbourne framework consists of 3 phases: (1) detection of health disparities and identification of vulnerable populations, (2) understanding why disparities exist, and (3) reducing or eliminating disparities through interventions. We focused on the first 2 phases and mapped the identified drivers of nonprescription antibiotic use onto the key domains of the Kilbourne conceptual framework: patient, healthcare system, and clinical encounter factors. We also conducted brief field research to explore anecdotal reports regarding availability of nonprescription antibiotics in our community. Results: We found 8 studies addressing factors related to nonprescription antibiotic use in the United States. These studies were primarily qualitative and included Spanish-speaking Hispanic and Latino immigrants. Figure 1 shows the proposed factors that may directly or indirectly predict nonprescription antibiotic use. Key potential factors are individual factors, psychosocial factors, resources, healthcare system factors, and clinical-encounter factors. For example, patients with inadequate health literacy may have poor access to care because of difficulty finding providers and choosing or navigating insurance plans; thus, they may be at risk for nonprescription use. At the same time, patients with inadequate health literacy may be at risk for using nonprescription antibiotics for a viral infection because of difficulty understanding medication labels or package inserts. The relevance of resources (availability) to nonprescription antibiotic use was supported by our research team’s purchase of amoxicillin, tetracycline, and metronidazole without prescriptions from a flea market in Houston, Texas. Conclusions: The Kilbourne conceptual framework provides a strong, comprehensive basis for research and intervention in the challenging problem of nonprescription antibiotic use. Ongoing research will test the proposed relationships between patient, healthcare system, and clinical-encounter factors and nonprescription antibiotic use outcomes. We are conducting a survey among both indigent and insured patient populations to identify the relative importance of these factors and to validate our proposed conceptual framework of nonprescription antibiotic use.
Funding: This project was supported by grant number R01HS026901 from the Agency.
Disclosures: None
Creating an Outpatient-Specific Antibiogram to Guide Treatment for Urinary Tract Infections
- Larissa Grigoryan, Melanie Goebel, Samuel Willis, Lisa Danek, Jennifer Matas, Neal Kachalia, Anna Katta, Kenneth Muldrew, Mohammad Zare, Forrest Hudson, Robert Atmar, Barbara Trautner
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s182-s183
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: Outpatients with uncomplicated urinary tract infections (UTIs) are often treated empirically without culture, whereas urine cultures are typically collected from patients with complicated UTI. Susceptibilities for fosfomycin (a first-line agent) are not routinely performed or reported in the antibiogram. Understanding the prevalence of antibiotic resistance for UTI is critical for empiric treatment and antibiotic stewardship in primary care. Methods: We developed a UTI-focused antibiogram from a prospective sample of outpatients (women and men) with UTIs from 2 public family medicine clinics in an urban area with a diverse, international population (November 2018 to present). During the study period, providers ordered a urine culture for any adult patient presenting with UTI symptoms, including uncomplicated and complicated infections. We estimated the prevalence of resistance to UTI-relevant antibiotics in the overall study population and compared it between patients born in the United States and other countries. Results: We collected 678 urine cultures from 644 unique patients (79% female). Of these cultures, 158 (23.3%) had no growth, 330 (48.7%) grew mixed urogenital flora, and 190 (28.0%) were positive (>10,000 CFU/mL). Patients with positive cultures were mostly female (88.2%), and their mean age was 46.6 ± 14.8 years. Among patients with positive cultures, 42.7% were born in the United States and 57.3% were born Mexico or Central America. Escherichia coli was the most commonly isolated organism (Fig. 1). Susceptibility results for E. coli and all gram-negative organisms combined are presented in Fig. 2. Susceptibility of uropathogens to TMP-SMX was significantly higher in patients born in the United States compared to patients from Mexico or Central America (82% vs 61%; P = .03). Susceptibility to ciprofloxacin was similar in patients born in the United States and other countries (79% vs 72%; P = .50). Of 77 E. coli isolates, 11 (14%) were positive for extended-spectrum β-lactamase production, including 8 isolates from patients whose country of origin was Mexico or a Central American country. Conclusions: More than 20% of outpatients presenting with UTI symptoms had a negative urine culture. Among outpatients with uncomplicated and complicated UTI, uropathogens had a high prevalence of resistance to ciprofloxacin and TMP-SMX, but susceptibility to fosfomycin (restricted in our system) was 100%. Resistance rates for TMP-SMX were higher in patients from Mexico and Central America. Our findings question whether TMP-SMX should remain a first-line agent in US primary-care settings.
Funding: This project was supported by the National Institute of Allergy and Infectious Diseases of the National Institutes of Health under Award Number UM1AI104681.
Disclosures: None
Effectiveness of Stewardship Intervention for Urinary Tract Infections in Primary Care: A Difference in Differences Study
- Larissa Grigoryan, George Germanos, Roger Zoorob, Mohamad Sidani, Haijun Wang, Mohammad Zare, Melanie Goebel, Barbara Trautner
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s515-s516
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Adherence to 2011 Infectious Diseases Society of America (IDSA) guidelines for urinary tract infections (UTIs) remains low in primary care. Fluoroquinolones are commonly prescribed to treat uncomplicated cystitis, and most antibiotic prescriptions have durations that exceed current recommendations. We performed a difference-in-differences study to assess the effectiveness of a stewardship intervention in a family medicine clinic at an academic outpatient center from August 2016 to March 2019. During our intervention period, the FDA released 2 additional warnings about the side effects of fluoroquinolones. Methods: The study had 2 sites (intervention and comparison) and 3 periods: baseline, before the intervention, and the intervention. During the first 2 years, we obtained baseline data and performed interviews (preintervention period) exploring provider prescribing decisions for cystitis at both sites. During the intervention period at the intervention site only, we presented an educational lecture including an overview of the IDSA guidelines, definitions for various UTI syndromes and actual clinical examples, and instruction on use of a decision aid. During the audit and feedback phase, providers were contacted once per month in person or by phone to provide follow-up on whether their treatment decision adhered to the IDSA guidelines. We performed a log-binomial regression analysis of the primary outcome, adherence to the IDSA guidelines for management of uncomplicated cystitis, both to antibiotic choice and duration of therapy. Results: We performed 156 audit-and-feedback sessions with 13 providers during the intervention period (March 2018–2019). Patients in both sites were similar in terms of age and Charlson comorbidity index. Adherence to the guidelines for antibiotic choice and duration increased in the intervention period at both sites (Fig. 1). The treatment of cystitis in the intervention period of the intervention site was 11.5 times (95% CI, 6.1–21.6) as likely to be guideline-adherent as the treatment in the baseline period of the comparison site (Fig. 2). Conclusions: Adherence to IDSA guidelines for the choice of antibiotic and duration increased in both intervention and comparison sites. Even though the intervention site started with higher compliance, improvement was also greater in the intervention site. FDA warnings about the side effects of fluoroquinolones released during the intervention period may have contributed to the avoidance of fluoroquinolones at both sites. Our intervention was effective at improving antibiotic choice and duration, so our future plans include incorporating our decision-support algorithm into the electronic medical record.
Funding: This study was supported by Zambon Pharmaceuticals.
Disclosures: None
Teamwork and safety climate affect antimicrobial stewardship for asymptomatic bacteriuria
- Dimitri M. Drekonja, Larissa Grigoryan, Paola Lichtenberger, Christopher J. Graber, Payal K. Patel, John N. Van, Laura M. Dillon, Yiqun Wang, Timothy P. Gauthier, Steve W. Wiseman, Bhavarth S. Shukla, Aanand D. Naik, Sylvia J. Hysong, Jennifer R. Kramer, Barbara W. Trautner
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 40 / Issue 9 / September 2019
- Published online by Cambridge University Press:
- 24 July 2019, pp. 963-967
- Print publication:
- September 2019
-
- Article
- Export citation
-
Objective:
In preparation for a multisite antibiotic stewardship intervention, we assessed knowledge and attitudes toward management of asymptomatic bacteriuria (ASB) plus teamwork and safety climate among providers, nurses, and clinical nurse assistants (CNAs).
Design:Prospective surveys during January–June 2018.
Setting:All acute and long-term care units of 4 Veterans’ Affairs facilities.
Methods:The survey instrument included 2 previously tested subcomponents: the Kicking CAUTI survey (ASB knowledge and attitudes) and the Safety Attitudes Questionnaire (SAQ).
Results:A total of 534 surveys were completed, with an overall response rate of 65%. Cognitive biases impacting management of ASB were identified. For example, providers presented with a case scenario of an asymptomatic patient with a positive urine culture were more likely to give antibiotics if the organism was resistant to antibiotics. Additionally, more than 80% of both nurses and CNAs indicated that foul smell is an appropriate indication for a urine culture. We found significant interprofessional differences in teamwork and safety climate (defined as attitudes about issues relevant to patient safety), with CNAs having highest scores and resident physicians having the lowest scores on self-reported perceptions of teamwork and safety climates (P < .001). Among providers, higher safety-climate scores were significantly associated with appropriate risk perceptions related to ASB, whereas social norms concerning ASB management were correlated with higher teamwork climate ratings.
Conclusions:Our survey revealed substantial misunderstanding regarding management of ASB among providers, nurses, and CNAs. Educating and empowering these professionals to discourage unnecessary urine culturing and inappropriate antibiotic use will be key components of antibiotic stewardship efforts.











